
In the mountains on earth and on the surface of mars water might always be in the solid state. Paul Morgan gathering water for dinner at 18,000 feet, 5,500 m, on the first ascent of Sierra Nevada de Lagunas Bravas in Chile.
Sitting around the old magnesium fire.
Paul Doherty and Eric Muller
Camping
First Things first
Oxygen
If you try to breathe Mars air you would pass out in 15 seconds due to lack of oxygen. So, campers would need to carry their own oxygen, O2. The movie "Total Recall" gives you an idea of what would happen to you if you tried to breathe Mars atmosphere.
Mars' atmosphere is 95% CO2 and 3% N2 and 2% Ar.
Demonstration. Put dry ice into a fish tank and let the tank fill with carbon dioxide. Gently waft some of the CO2 toward your nose, smell it to find out what Mars smells like. Stick your tongue into the tank and taste the Co2. Wow! be prepared for a sharp taste like that from a carbonated beverage.(We'll use the tank of CO2 again in other experiments below so keep it around.)
However, when water is added to the surface of Mars it releases oxygen, so all you have to do is pee on the surface to generate O2. Viking landers discovered that the surface contained iron superoxides, when Viking added water to the soil oxygen was released. Scientists from NASA AMES research laboratories recently found the same kind of oxidizing soil in the Atacama desert of Chile.
Demo, Get a platinum coated plastic contact lens cleaner. Drop it into a test tube of hydrogen peroxide (20%) and oxygen will bubble out. H2O2 goes to H2o + O. This models the production od oxygen by mars soil when it is wetted.
If there is water on Mars then solar power can be used to split the water, H2O, into hydrogen and oxygen.
Water
The MER spacecraft are searching for signs of water on Mars. Spacecraft show that the soil near the surface on Mars near the equator is only 3% water. The polar caps do contain a lot of water ice. The Mars Express spacecraft in orbit carries radar which is looking for water ice beneath the surface of Mars. So until they find water, top up your water bottle before you go.

In the mountains on earth and on the surface of mars water
might always be in the solid state. Paul Morgan gathering water for
dinner at 18,000 feet, 5,500 m, on the first ascent of Sierra Nevada
de Lagunas Bravas in Chile.
Fire
Most things won't burn on mars, after all, the main ingredient in the atmosphere, carbon dioxide or CO2, is used in fire extinguishers on Earth.
Demo, make a fish tank full of CO2 gas. Drop dry ice into warm water in a pitcher to create a cloud, pour the cloud into the tank and notice how it floats as a stratus cloud layer above the CO2 in the tank. Carbon dioxide is heavier than air and so it sinks into the tank and stays there. Lower a candle into the tank and then a lighter and watch them extinguish. Notice in particular how the flame from the lighter leaves the top of the CO2 and enters the air. Try a camping stove it also goes out as it is lowered into the tank.
However, metals will burn in a CO2 atmosphere.
Demo: Eric lowers a burning magnesium ribbon into the carbon dioxide atmosphere in the tank, it continues to burn. Magnesium can tear apart the carbon dioxide, burn with the oxygen to make magnesium oxide, and leave the carbon behind The magnesium ribbon becomes white, magnesium oxide, with black spots from the carbon.
Place magnesium powder into a hollow carved into two blocks of dry ice. Insert a fuse of magnesium ribbon into the powder and light the fuse. Put one block on top of the other to seal the magnesium from air. The magnesium burns inside the CO2 producing a beautiful glow. Open the blocks when the fire is out and notice the white magnesium oxide and the black carbon left behind.
Cooking
Cooking is difficult, the pressure is so low on Mars that water boils at 0°C. You would definitely need to use a pressure cooker.
Air Pressure on Mars, is less than 1/100 of the air pressure on earth.
Demonstration, A steel rod 1 inch square, 2.5 cm square, and 5 feet or 1.3 m long will exert the same pressure as the earth's atmosphere. To feel Martian pressure cut the length of the rod to 1/100 of this length or 1.3 cm. A Venus atmosphere rod would be 500 feet high, 160 m, and weigh 1470 pounds, nearly a ton.


Left, a steel Bar 1.32 m long and 1 inch square exerts the same
pressure as the earth's atmosphere, it weighs 14.7 pounds. (101
Kpascals.) A steel Mars atmosphere bars is only 1.32 cm
high.
The unit of atmospheric pressure on earth is the Bar, on mars it is of course the mars bar. Don't worry if you can't cook on Mars you can always eat... mars bars!
Demo, Show the candy bar.
At Mars pressure, water boils at 0 °C, you can show this using a vacuum chamber or a syringe.
So to cook on Mars you'll need a pressure cooker. Spaghetti cooked in a carbon dioxide martian atmosphere pressure cooker woud be spaghetti carbonara.
Altitude Illness
On earth, to deal with illness caused by low atmospheric pressure, climbers get into a Gamow Bag which can be pumped up to high pressure. It's rather claustrophobic, and you do look like a hot dog, but it can save your life. Perhaps the first Mars team will live in a pressurized fabric dwelling.
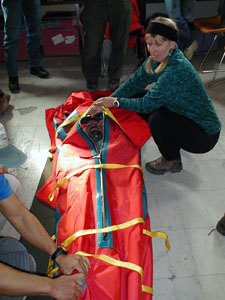

Left, a person inside an open Gamow bag, right, an inflated
Gamow bag. Click images to enlarge.
Gravity
Your backpack would weigh less under the 1/3 g
gravity of Mars.
But you'll need to carry more stuff.
Temperature
The temperature will never get below -210 °F, -130 °C,since at that point the Martian air freezes solid and releases energy as it does so. But even a warm night gets down to -100 °F, as cold as the coldest winter nights in Antarctica. So you'll need a thick sleeping bag.
Demo show a -40 degree sleeping bag(-40 is the same temperature in Fahrenheit or Celsius.) Then a -80 F sleeping bag (made by putting an overbag over the -40 bag.) Notice how thick the bag is.

Packed in a tent like sardines. -40°F outside, Sierra Nevada,
Chile.
You'll need a wind breaker since the winds are fast, but the air is thin, so it will exert less force.
However complete thermal protection from the cold will be required, here is a picture of Noel Wanner on the summit of Mt. Erebus in Antarctica, at the time of this photo it was -20 C with 20 mph winds. When Noel removed his face mask for a minute his nose turned white with incipient frostbite.

Noel Wanner dressed for the summit of a cold Antarctic
mountain. No flesh is showing. He lowered his hood for the
photograph.

Paul Doherty climbing Ojos de Salado, -30 C and 30 mph winds.
Working hard going uphill at 6000 m elevation, just below 20,000
feet, and still cold.
Navigation
A compass will not work on Mars. Neither will GPS.
There is a South Pole Star on Mars, Kappa Velorum, magnitude 2.46 just 2 degrees from the actual pole. Polaris, the north pole star on earth is brighter, and closer to the pole, less than a degree from the north celestial pole. There is no north pole star on Mars just as there is no south pole star on earth.
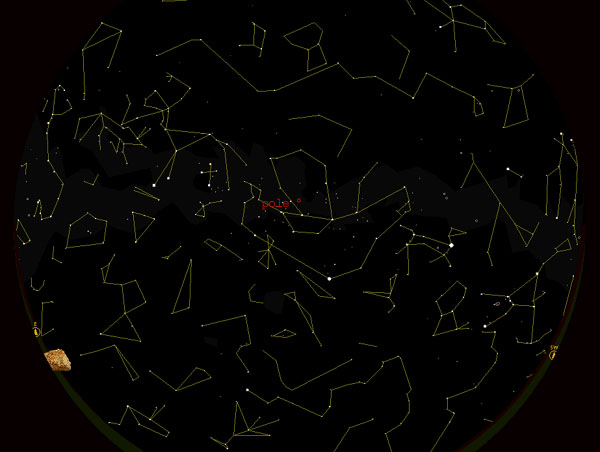
The south pole star on Mars is Kappa Velorum, just to the left
of the pole in this image. Click to enlarge.
The thermal emission spectrometer sent back data on the temperature and composition of rocks around the spirit lander site.

In this image the TES data is placed over a lander pancam
image. The colors indicate the temperatures of the surface. In this
daytime image the rocks are shown in blue representing cooler
temperatures (up to 10 °C cooler than average, while dust is red
and is up to 10°C warmer.
TES also took spectra of the infrared radiation emitted by the surroundings. In the following image the spectra is shown in yellow. Spectra of the site from orbit are shown in red.
Notice the middle wavelength here is 10 micrometers, over ten times the wavelength of red light, and ten times smaller than the diameter of a human hair. The dips in the spectra represent absorption by different minerals. This data shows the presence of silicates, and carbonates in the rock as well as, carbon dioxide in the atmosphere and even water.

Images were sent back of the sundial on the spirit lander, it is also
a color calibration target. Notice how scattering of light from the
sky fills in the shadow of the gnomon. Skylight is about equal to the
brightness of direct sunlight on Mars, so the shadow is darker by
about one f-stop, a factor of two in photographic exposure
terms.
|
Scientific Explorations with Paul Doherty |
|
25 Dec 2003 |