Blue light photons have higher energy than red
Introduction
Light emitting diodes, LEDs, convert the energy change of one
electron into one light photon. Slowly increase the energy of the
electrons by increasing the voltage across the LEDs and you will see
that the red LED comes on at the lowest voltage then the green and
finally the blue at the highest voltage.
Material
Assembly
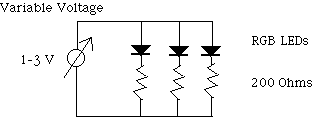
Connect the LED’s in series with the 200 ohm resistors.
Connect the LED’s and resistors in parallel then connect them to
the variable voltage supply.

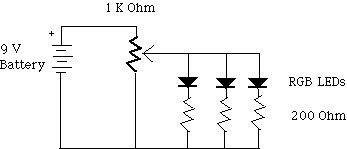
Or, if you wish to use the 9 volt battery, then build a voltage
divider. Connect the 1 kW
potentiometer to the 9 volt battery, then connect the diodes with
their series resistors in from the output of the potentiometer to
ground.
To Do and Notice
Slowly increase the voltage across the LED’s.
Notice that first the red LED comes on, then the green and finally
the blue.
Then repeat the process of raising the voltage.
When you first see light in the red LED use the voltmeter to measure
the voltage across the LED.
When the green LED first comes on, measure the voltage across its
leads.
When the blue LED comes on, measure the voltage across its leads.
What’s Going On?
A light emitting diode changes the electrical energy of one electron
into the light energy of one photon. Thus lower energy photons of red
light are produced when the electrons drop through smaller voltage
differences while higher energy blue photons require higher voltage
differences. It is interesting to see the lights come on in order,
first red then green then blue
Incandescent bulbs convert the energy change of one electron as it crosses a resistance into more than, or fewer than one photon.
More detail
The negative electrons have high energy at the negative terminal of the battery. In the LED they enter the N-type semiconductor and drop all of their energy at the junction with a P-type semiconductor as they fall into holes. This energy is emitted as electromagnetic radiation, visible light in LEDs. The electrons then return to the positive terminal of the battery. In an incandescent bulb the electrons drop their energy gradually as they travel through the filament. Their electrical energy is converted to thermal energy. This thermal energy is then converted into electromagnetic radiation.
Math Root
The energy, E, in joules, of a photon is proportional to its frequency, f, in hertz.
Where h is the constant of proportionality known
as Planck's constant.
h = 6.6 x 10^-34 J s
Red light has a frequency near 500 THz, (500
Terahertz, 500 x 10^12 Hz)
Green near 600 THz
Blue near 750 THz
So the energy of a red photon is E = 6.6 x 10^-34 x 500 x 10^12 = 3.3 x 10^-19 J
and the blue E = 5 x 10^-19 J.
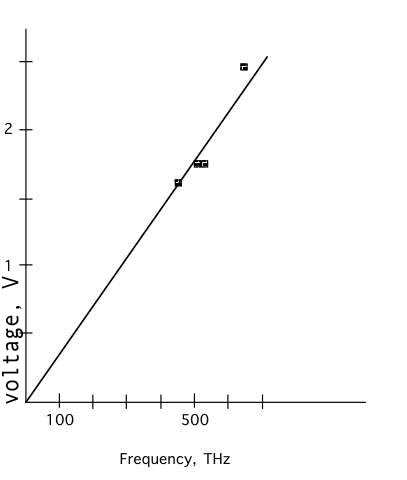
You can plot the Voltage for each LED versus the frequency
Recall the equation relating the energy of a photon, E, to its frequency, f, and the equation relating the energy of an electron of charge q, to its voltage, V.:
E = hf and E = qV
an LED makes these equal so the line represents
qV = hf
the slope of the voltage versus frequency line is then
V = h/q f
The slope is Planck's constant divided by the charge on an electron q which is 1.6 x 10^-19 C
The slope should be 6.6 x 10^-34/1.6 x 10^-19 or about 4 * 10^-15 Js/C or 4 V/GHz
(A GHz is 1000 THz)
The slope of our line is about 3 Volts per GHz
Not a bad measurement for under $10.
Etc
You can look at the light produced by the LEDs through a diffraction grating. You will see that the deflection of the image of the lamp is proportional to the wavelength of the light, the longer wavelength red is deflected more than the shorter wavelength blue. A commercial spectroscope can be calibrated in light frequency or energy so that the frequency of the light from each LED can be read off the spectroscope.
The spectroscope will give the wavelength of the light not its frequency. To convert wavelength, L, to frequency, f, you will need to calculate c = fL, where c is the speed of light 3 x 10^8 m/s.
Scientific Explorations
with Paul Doherty 22 May 2000