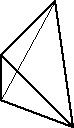
A tetrahedron. In a silicate there would be an oxygen at every vertex and a silicon in the center.
I love rocks.
While everyone else is looking at birds or butterflies or flowers, I'm looking at rocks.
Paul Doherty
Here is a physicist's view of rocks.
There are three types of rocks:
Igneous which form from a melt.
Sedimentary which are rocks which form from sediments or from solution.
Metamorphic which are formed from other rocks by recrystalization under temperature and/or pressure.
Igneous Rocks
Igneous rocks are all made when a liquid cools and solidifies. The most common igneous rocks are made of silicate minerals. (90% of the earth's crust is made of silicate minerals.)
Silicate minerals are made of silicon and oxygen tetrahedra.
Silicate minerals are formed from tetrahedrons with a silicon atom at the center of each tetrahedron and an oxygen at each vertex. The silicon has a +4 valence while the oxygens are -2 resulting in a silicate which is SiO4-4.

The silicon having lost four electrons is much smaller than the oxygens which have gained two electrons. The silicon is almost exactly the right size to fit in the hole left in the middle when 4 oxygens make a tetrahedron.
The silicon bonds to the oxygens with bonds that are 50% covalent and 50% ionic.
The silicate tetrahedrons can bond together with ions between each silicate.
For example if +4 silicons are between every pair of silicate tetrahedrons in a 3-dimensional arrangement then the clear mineral quartz is formed with a chemical equation SiO2.
Silicate tetrahedra can bond together in 3 dimensions with some tetrahedra containing aluminum instead of silicon. Since aluminum has a valence of +3 the mineral will require other positive ions such as calcium, Ca+2, Sodium, Na+1 or potassium K+1 or a mixture of these ions between the tetrahedra. This makes an opaque silicate mineral, feldspar.

Silicates can bond together in planes with many interstitial atoms to form flat plate mica.
Combine quartz, feldspar and mica to make a rock known as granite.
A good web site containing mineral descriptions is the mineral gallery.
Opal has a composition of SiO2 +nH2O but it is amorphous and not crystalline it is not a silicate mineral.
When water freezes it creates the mineral ice. Ice is a common mineral on the cold moons such as Titan in the outer solar system. Ice fresh from the earth freezer has a hardness of 1.5 on the mohs scale, ice at -70 C in Antarctica or on a cold moon has hardness 6 and can erode feldspar.
Igneous rocks 2
When rocks cool from a melt crystals form.
The longer the cooling time the larger the crystals.
Obsidian glass is a rapidly cooled igneous silicate rock with no crystals.
Granite with large crystals visible to the unaided eye is a slowly cooled igneous rock.
Rocks are composed of minerals with different chemistries. The main division of chemistry is by iron content. Rocks rich in silica and aluminum are called felsic and are light colored like granite and rhyolite while rocks rich in iron and manganese are called mafic and are dark colored like basalt and gabbro.
Rhyolite has small crystals while granite has large crystals.
Basalt has small crystals while gabbro has large crystals.
Sedimentary rocks
Sedimentary rocks made from sediments are classified by grain size.
Grain size Sediment rock > 2 mm gravel conglomerate 0.06 to 2 mm sand sandstone 0.002 to .06 mm silt siltstone <0.002 mm clay shale
Conglomerate sedimentary rock contains gravel that is visible to the unaided eye.

Metamorphic rocks
Metamorphic rocks are rocks which have been transformed by heat, pressure or reactions with water.
Limestone made of calcium carbonate grows larger crystals to become marble.
Sandstone made of crystals of quartz cemented together by amorphous silicate cement metamorphoses into a rock known as quartzite where the silicates of the amorphous cement become part of adjacent crystal grains.
Shale metamorphoses to slate then to phyllite to schist and finally to gneiss.
Slate is black and harder than shale, phyllite has a shiny surface schist has abundant mica flakes a couple of millimeters across and gneiss is separated into dark and light bands.
A website on metamorphic rocks with photos.
|
Scientific Explorations with Paul Doherty |
|
15 April 2005 |