
Tree rings record past climate.
Weather Past Workshop
Thermometer records
Human memory is a fallible recorder of climate, however there are records of thermometer readings for over 100 years.
Tree Rings

Tree rings record past climate.
Trees also record climate in their yearly growth rings, they record a complex mix of temperature and rainfall. Good growth years have wide growth rings and poor years have narrow rings. In the American west bristlecone pine growth rings record the climate for the past 10,000 years.
Sediment cores.
Sediments get washed into the ocean (and lakes) where they settle to the bottom.
Pour alternating layers of dark and light sand into a transparent sided box to show sediment layering.
Notice what was discovered by Steno four hundred years ago, the oldest layers are on the bottom.
When scientists drill down into the sediments and sedimentary rock they are going down and also back in time.

Stephanie Chasteen makes a core of the cake.

Examining the core before slicing it in half. Notice the close up in
the right hand monitor.
We can model the sediment coring process using a layer cake and the thin plastic tubes that are used to protect fluorescent lightbulbs. Take a 50 cm (20 inch) long piece of the thin plastic tube and slowly twist it while pushing it down into a layer cake. Then slowly pull it out of the cake to reveal a cake-sediment core. You can then cut off the piece of the core that contains the cake and using scissors and a knife cut it in half along its depth. Then open the two sections like a book to reveal a clear set of sedimentary layers. This is exactly what they do with real sediment cores.
Oxygen Isotopes
Oxygen comes in two different isotopes, the lighter oxygen 16 and the heavier oxygen 18. In ocean water at a given temperature water molecules containing the lighter isotope of oxygen, H2O, move faster than those containing the heavier oxygen isotope. These faster moving molecules escape from the water more rapidly, they evaporate, leaving the heavier isotopes behind in the liquid water. BY measuring the oxygen isotopes in ice scientists can determine the temperature of the water that evaporated to form the snow that fell to make the ice.
As a model of this process, take a shallow plastic tray about 20 cm wide 40 cm long and 5 cm deep. Place 8 golf balls representing water molecules containing the heavier oxygen 18 isotopes, and 8 ping pong balls representing water containing the lighter oxygen isotopes into the tray. Gently shake the tray to simulate the motion of molecules in warm water. Notice how the lighter ping pong balls evaporate much more rapidly than the heavier golf balls. This represents the oxygen isotope separation that occurs in nature.
Ice Cores
Ice Balloons, Fill a balloon with water and place it in the freezer for 24 hours. Take it out of the freezer and remove the balloon from a ball of ice. Look at the ice. Notice the air bubbles trapped in the ice.

Wash off the ice balloon and observe the bubbles inside the
balloon.
The ice freezes at the outside first. The resulting shell of ice traps the remaining water. Water from the tap contains dissolved air. As the water freezes it crystallizes into pure water ice forcing the dissolved air to move into the remaining liquid water. At some point the dissolved air comes out of solution making an air bubble. This air bubble is trapped in the ice. Snow as it falls on a glacier contains a great deal of trapped air. As the snow is buried under more snow that falls later the snow recrystallizes into larger crystals of ice forcing the air into bubbles that are trapped in the ice. Thus glacier ice in ice cores traps air from the atmosphere.
Put it all together
Ice cores preserve yearly layers which can be counted to determine the age of the ice core. They contain bubbles of dissolved gas which can be analyzed to find the composition of the atmosphere in the past, and they contain isotopes of oxygen which can be used to determine the temperature of the ocean water which evaporated to form the snow that made the ice. Ice cores thus preserve a record of atmospheric composition and temperature. As of 2006 ice cores had been collected stretching back over 400,000 years.
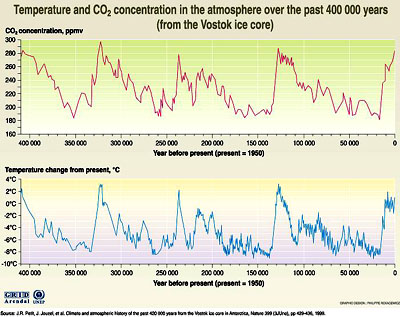
The record of temperature and CO2 concentration from the Vostok ice
core for the past 400,000 years.
Notice that the concentration of CO2 is strongly correlated with temperature. The ice core traps air from fifty years ago, since the top of this ice core trapped its last air the concentration of CO2 in the atmosphere has risen to 380 ppm. shooting way off the top of this graph. Scientists are worried about what will happen to the temperature of the earth's atmosphere.
|
Scientific Explorations with Paul Doherty |
|
21 November 2006 |